Overview
This article delves into the techniques and applications of mastering oxalic acid pKa, underscoring its critical role in acid-base chemistry and pharmaceutical contexts. Understanding the pKa values of oxalic acid, specifically 1.25 and 4.27, is essential for predicting its behavior in chemical reactions and drug formulations. The discussion is bolstered by various measurement techniques, including:
- Potentiometric titration
- UV-Vis spectrophotometry
These techniques enhance the accuracy of determining these values. By comprehending these techniques, researchers can ensure precision in their work, ultimately leading to improved outcomes in both laboratory and pharmaceutical settings.
Introduction
Understanding the nuances of oxalic acid pKa is pivotal in both academic and industrial chemistry. This concept serves as a cornerstone for predicting the behavior of this versatile compound in various environments. By delving into the significance of pKa values, readers will explore how these measurements influence:
- Solubility
- Drug absorption
- Effectiveness of chemical reactions
However, a challenge arises: how do these seemingly abstract numbers translate into real-world applications that can impact everything from pharmaceuticals to industrial processes? This exploration is essential for grasping the broader implications of pKa in various fields.
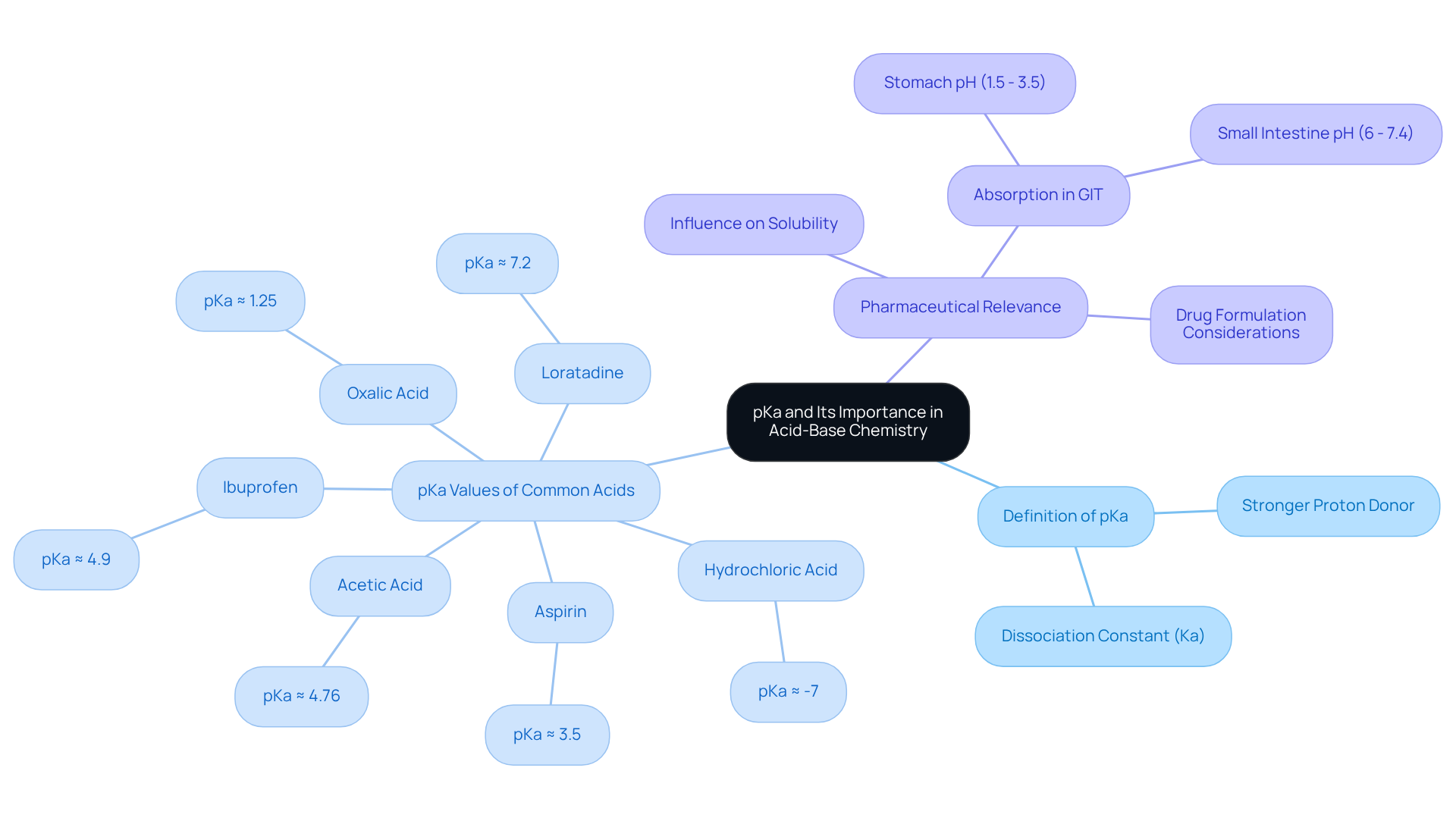
Define pKa and Its Importance in Acid-Base Chemistry
The pKa of oxalic acid, or the dissociation constant, serves as a critical metric for quantifying the strength of a compound in solution. It is defined as the negative logarithm of the dissociation constant (Ka). A lower oxalic acid pKa value indicates a stronger proton donor, reflecting a greater degree of dissociation in solution. This understanding is essential for predicting the behavior of acids, such as oxalic acid pKa, in various chemical reactions, particularly in acid-base equilibria. For example:
- Hydrochloric acid, a strong acid, has a pKa of approximately -7.
- The oxalic acid pKa is around 1.25.
- Weaker acids like acetic acid exhibit a pKa around 4.76.
In pharmaceutical contexts, the oxalic acid pKa of a drug plays a pivotal role as it influences its solubility and absorption within the gastrointestinal tract (GIT), which presents varying pH levels. The stomach's pH typically ranges from 1.5 to 3.5, while the small intestine's pH varies between 6 and 7.4. For instance:
- Ibuprofen, with a pKa of 4.9, is less non-ionized in the acidic environment of the stomach compared to aspirin, which has an oxalic acid pKa of about 3.5, thus allowing for better absorption of aspirin.
- Loratadine, considering the oxalic acid pKa of approximately 7.2, remains more non-ionized in the small intestine, enhancing its absorption potential.
Recent studies emphasize the significance of oxalic acid pKa in drug formulation, underscoring the necessity for drug developers to consider oxalic acid pKa and pH-dependent absorption profiles during the design of medications. As noted by Mohanned Jallad, 'Drug developers and clinicians consider the pKa and pH-dependent absorption profiles of drugs when designing formulations and determining dosing regimens.' Understanding the of active pharmaceutical ingredients is fundamental for optimizing drug delivery and efficacy, marking it as a crucial factor in pharmaceutical research and development.
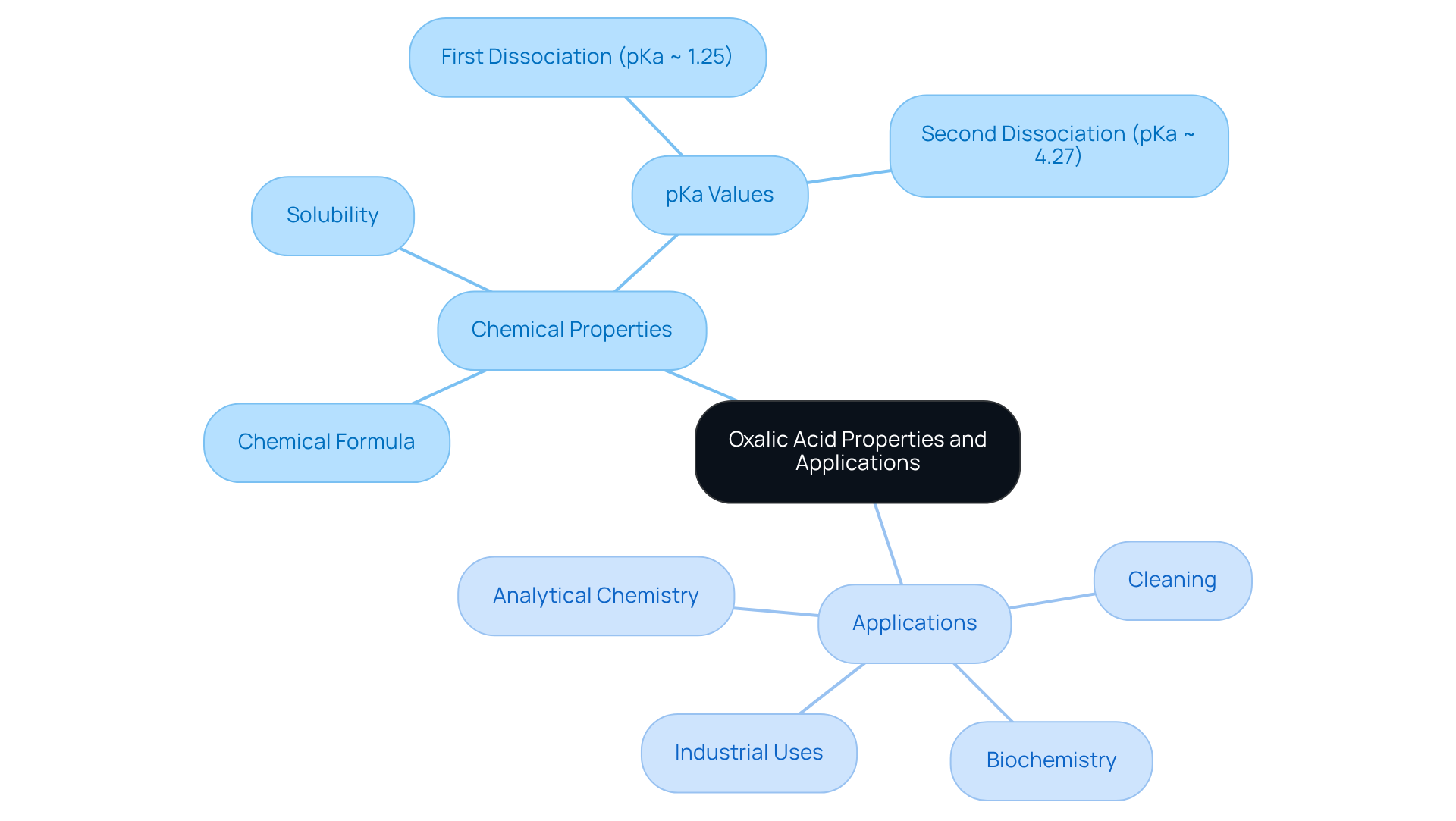
Explore the Properties of Oxalic Acid and Its Applications
Oxalic compound (C2H2O4) is a colorless, crystalline organic substance known for its high solubility in water. As a dicarboxylic compound, it contains two carboxyl groups (-COOH), which are responsible for its acidity. The compound exhibits two pKa values, which are referred to as the oxalic acid pKa: approximately 1.25 for the first dissociation and 4.27 for the second. This dual dissociation renders it a versatile reagent with various applications that are critical in multiple fields.
In analytical chemistry, oxalic compound serves as a primary standard for titrations, particularly in redox reactions, ensuring precision in measurements. As a cleaning agent, it effectively removes rust and stains from metals, thanks to its strong chelating properties. In biochemistry, it plays a significant role in metabolic processes and is involved in the formation of calcium oxalate crystals, which can have implications for kidney health. Furthermore, in industrial applications, it is utilized in the production of dyes and bleaches, and acts as a reducing agent in various chemical processes.
The multifaceted nature of oxalic compound underscores its importance across different sectors, highlighting the need for a thorough understanding of its properties and applications.
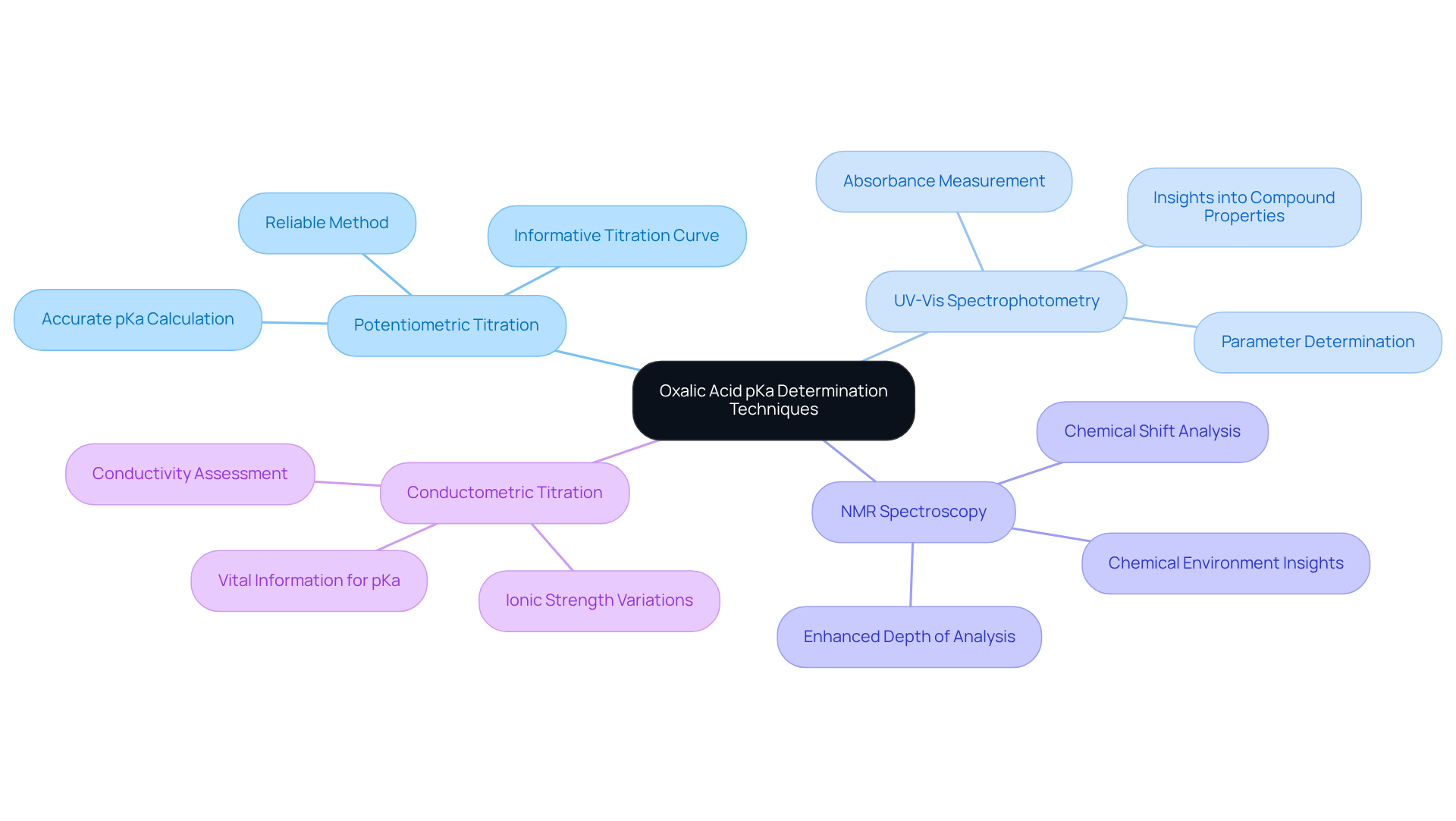
Detail Measurement Techniques for Determining Oxalic Acid pKa
Determining the oxalic acid pKa is essential for understanding its chemical behavior, and several techniques can effectively accomplish this task, each offering distinct advantages.
- Potentiometric Titration stands out as a reliable method, involving the titration of an organic compound with a strong base while meticulously measuring the pH. The pKa of oxalic acid can be accurately calculated from the inflection points observed on the titration curve, making this technique both precise and informative.
- UV-Vis Spectrophotometry provides another valuable approach. By measuring the absorbance of solutions at various pH levels, one can ascertain the parameters of oxalic acid pKa based on the alterations in absorbance, revealing critical insights into the compound's properties.
- NMR Spectroscopy further enriches our understanding by offering insights into the chemical environment of protons within the dicarboxylic compound. Through chemical shift analysis, researchers can determine the oxalic acid pKa values, which enhances the depth of analysis.
- Lastly, Conductometric Titration assesses the conductivity of the solution throughout the titration process. This method supplies vital information to determine oxalic acid pKa figures based on variations in ionic strength, rounding out the toolkit available for accurate pKa determination.
Each of these methods necessitates careful calibration and control of experimental conditions, underscoring the importance of precision in obtaining accurate results.
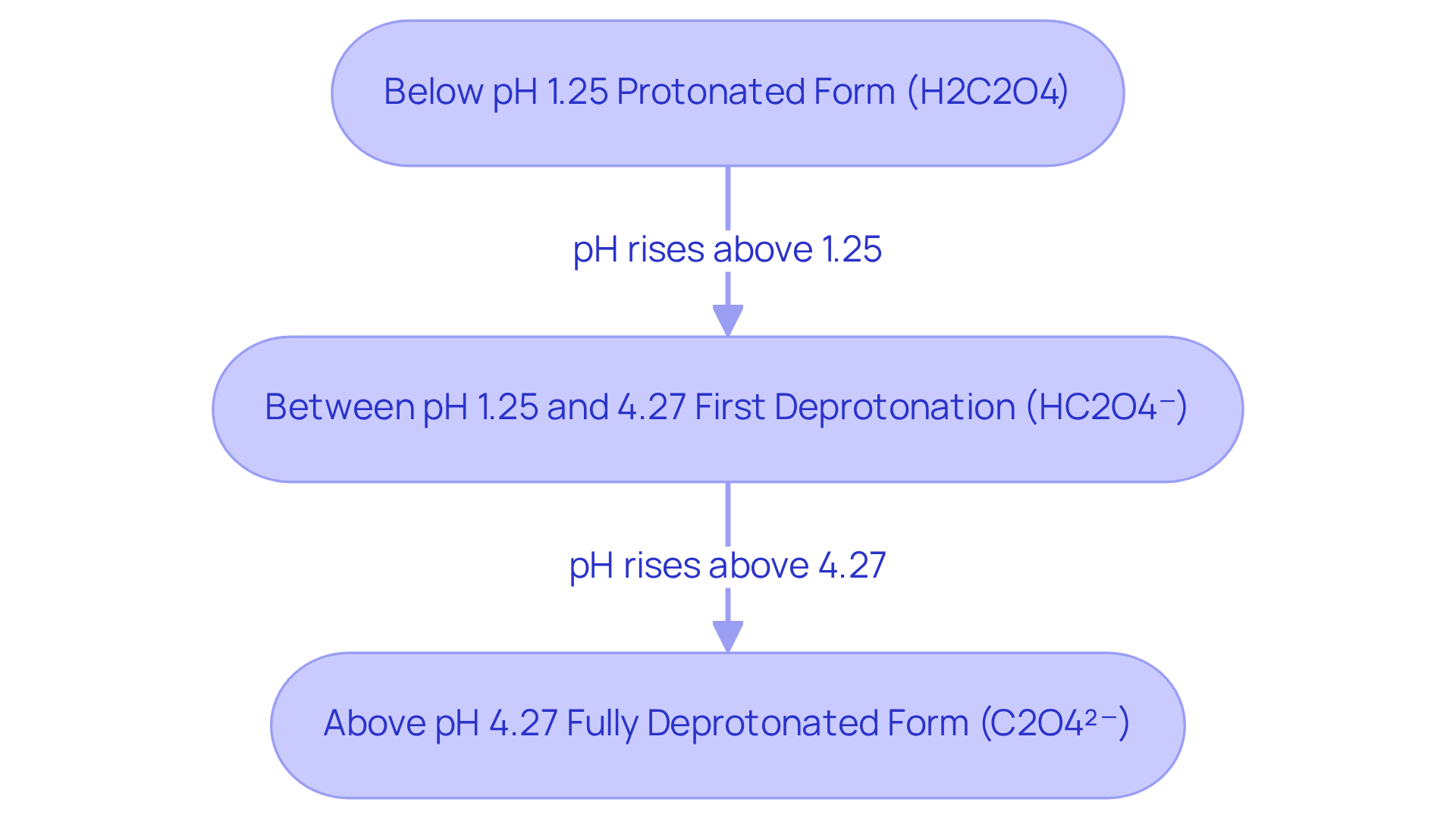
Interpret pKa Values and Their Implications in Practice
Understanding pKa measurements is essential for forecasting the behavior of dicarboxylic compounds in various chemical reactions. The two oxalic acid pKa values, 1.25 and 4.27, highlight its capacity to donate protons at different pH levels.
At pH values below the oxalic acid pKa of 1.25, oxalic acid predominantly exists in its protonated form (H2C2O4). As the pH rises beyond this threshold, it initiates the loss of its first proton, transitioning to HC2O4⁻.
Second Dissociation (oxalic acid pKa = 4.27): When the pH exceeds the oxalic acid pKa of 4.27, the second proton is released, resulting in the fully deprotonated form (C2O4²⁻).
Grasping these transitions is vital for applications such as buffer preparation, where maintaining a specific pH is crucial. Moreover, in drug formulation, an understanding of pKa is instrumental in predicting solubility and absorption characteristics, which ultimately influences the efficacy of pharmaceutical compounds.
Conclusion
Grasping the pKa of oxalic acid is essential for understanding its role in various chemical contexts, particularly in acid-base chemistry. The importance of pKa transcends mere numerical values; it acts as a crucial indicator of how oxalic acid behaves across different environments. With a pKa of approximately 1.25 for its first dissociation and 4.27 for the second, this dicarboxylic acid exhibits distinct proton-donating capabilities that are vital for predicting its interactions in laboratory and pharmaceutical settings.
This article explores the properties and applications of oxalic acid, showcasing its versatility as a reagent in analytical chemistry, a cleaning agent, and its significance in biochemistry and industrial processes. Techniques for accurately determining its pKa, such as potentiometric titration and UV-Vis spectrophotometry, are critical for researchers seeking to optimize its use in drug formulation and other applications. Understanding the transitions between its protonated and deprotonated forms based on pH not only facilitates buffer preparation but also enhances the efficacy of pharmaceutical compounds.
In conclusion, mastering the pKa of oxalic acid is more than an academic pursuit; it carries practical implications across multiple fields. As the importance of pKa in acid-base reactions becomes increasingly evident, it is crucial for chemists and drug developers to incorporate this knowledge into their work. By doing so, they can refine drug delivery systems, improve product formulations, and ultimately contribute to advancements in both scientific research and practical applications.




