Overview
The article presents ten essential applications of 0.22 micron syringe filters in laboratories, underscoring their vital role in ensuring sample purity across a range of contexts, including pharmaceutical manufacturing, environmental testing, and biotechnology. Evidence supports the assertion that these filters effectively eliminate microorganisms, thus enhancing the reliability of analyses and ensuring compliance with regulatory standards. This highlights their significance in maintaining safety and quality throughout laboratory processes. In summary, the adoption of 0.22 micron syringe filters is not merely beneficial but essential for upholding the integrity of scientific work.
Introduction
The precision and reliability of laboratory results are fundamentally dependent on the purity of the samples being analyzed. Among the various tools at our disposal, the 0.22 micron syringe filter emerges as an indispensable device for ensuring the effective elimination of contaminants such as bacteria and fungi from solutions. As the demand for stringent quality control in laboratories intensifies, it becomes essential to comprehend the diverse applications of these filters.
- What challenges do researchers encounter in maintaining sample integrity?
- How can the appropriate filtration techniques mitigate these risks?
Understanding these aspects is crucial for advancing laboratory practices and ensuring the highest standards of quality.
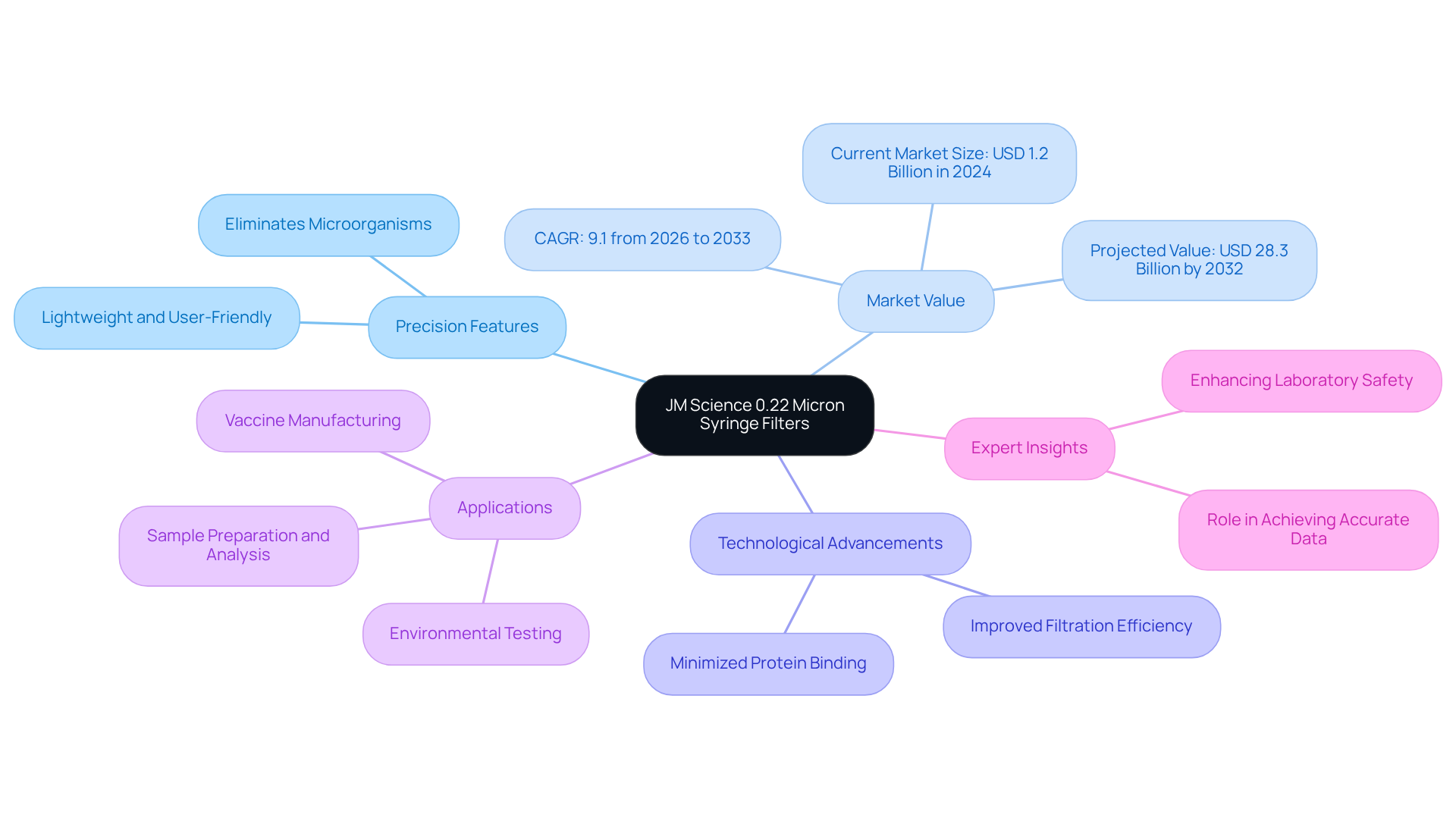
JM Science 0.22 Micron Syringe Filters: Precision for Laboratory Use
JM Science's syringe filter 0.22 micron membranes are designed for exceptional precision in laboratory applications, playing a pivotal role in ensuring sample purity. The syringe filter 0.22 micron effectively eliminates bacteria, fungi, and other microorganisms from solutions, guaranteeing sterility and reliability in various laboratory tasks, including sample preparation and analysis. Lightweight and user-friendly, they have become the preferred choice among researchers and technicians.
The is projected to expand significantly, with an estimated value of USD 28.3 billion by 2032, reflecting the increasing demand for accurate sample purification. Recent advancements in syringe membrane technology have further optimized their performance, emphasizing improved filtration efficiency and minimized protein binding. As laboratory professionals from MBP Inc. assert, "Syringe membranes play a crucial role in achieving accurate data, protecting equipment, and enhancing laboratory safety."
The practical applications of syringe filter 0.22 micron membranes extend to vaccine manufacturing, where it is vital to maintain sterility. Furthermore, they are utilized in environmental testing to ensure that samples are devoid of contaminants that could compromise analytical results. As the demand for precise sample purification continues to escalate, the syringe filter 0.22 micron plays an indispensable role in laboratory environments.
Pharmaceutical Applications: Ensuring Compliance with 0.22 Micron Filters
In the pharmaceutical sector, the use of a syringe filter 0.22 micron is essential for meeting stringent regulatory requirements. These devices play a pivotal role in sterilizing injectable solutions and other sensitive items, effectively eliminating contaminants that could jeopardize patient safety. By integrating small screens into their processes, pharmaceutical companies not only uphold the quality of their products but also ensure compliance with Good Manufacturing Practices (GMP). Notably, over 60% of pharmaceutical-grade applications rely on these systems, underscoring their critical role in quality preservation.
Regulatory experts emphasize that using a syringe filter 0.22 micron is vital for safeguarding against microbial contamination, thereby enhancing overall product safety and effectiveness. As Kate Douglass, President of Performance Strategies, LLC, aptly states, "The change you embrace today will improve your current sterile compounding practices and reduce patient risk tomorrow."
Leading pharmaceutical companies, such as Pfizer and Merck, have adopted these mechanisms as standard practice in their manufacturing processes, demonstrating their commitment to quality and compliance. As the pharmaceutical purification market is projected to reach USD 20.97 billion by 2029, the reliance on 0.22-micrometer sieves is expected to increase, further solidifying their importance in ensuring the safety and efficacy of injectable solutions.
To maintain compliance with GMP, pharmaceutical firms must routinely evaluate their filtration methods and incorporate syringe filter 0.22 micron membranes as a standard in their .
Environmental Testing: Utilizing 0.22 Micron Filters for Water Quality
Devices utilizing a syringe filter 0.22 micron play a crucial role in environmental testing, particularly in the evaluation of water quality. These instruments, such as the syringe filter 0.22 micron, are meticulously engineered to effectively from water samples, thereby ensuring the precision and reliability of microbiological analyses.
Environmental scientists emphasize that utilizing a syringe filter 0.22 micron significantly enhances the accuracy of data collected during environmental monitoring and public health assessments. Research has shown that employing a syringe filter 0.22 micron in water quality evaluations leads to a more precise identification of microbial pollutants, which is vital for safeguarding public health.
Additionally, the ability of 0.22-millimeter screens to obstruct airborne pollutants while allowing gases or vapors to pass underscores their importance in maintaining sample integrity. Consequently, laboratories that utilize these devices can provide essential information that informs regulatory compliance and environmental safety initiatives.
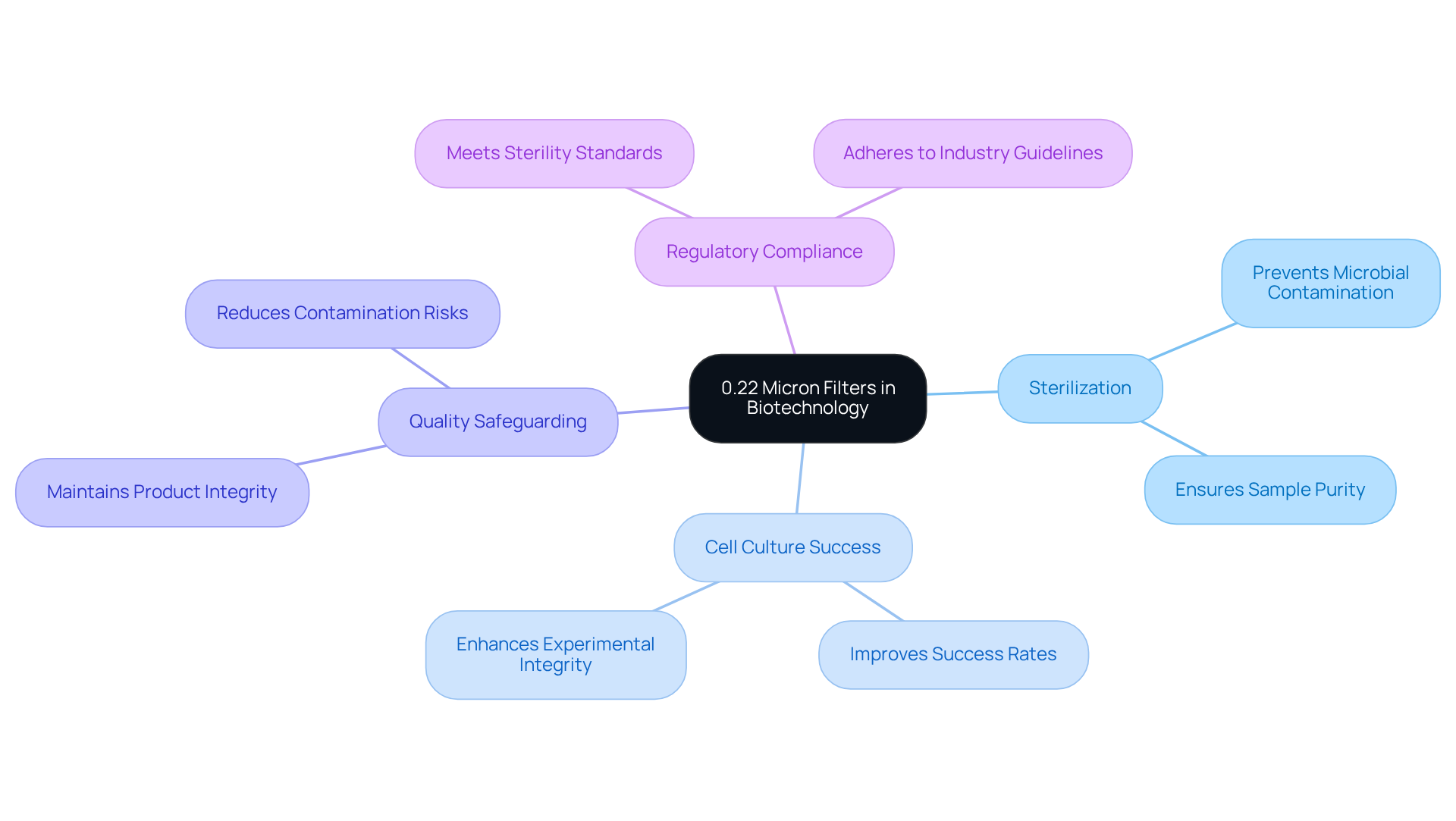
Biotechnology Applications: Sterilizing Solutions with 0.22 Micron Filters
In biotechnology, the use of a syringe filter 0.22 micron is indispensable for sterilizing solutions utilized in cell culture and protein purification. These devices effectively eliminate impurities, a crucial factor for the success of experiments and the advancement of biopharmaceuticals.
Employing 0.22 nanometer filtration enables researchers to uphold the sterility of their samples, significantly enhancing precision in their findings. Research indicates that utilizing these barriers can lead to improved cell culture success rates, as they prevent microbial contamination that could compromise experimental integrity.
Biotechnologists emphasize that integrating a syringe filter 0.22 micron into standard procedures not only safeguards quality but also complies with stringent regulatory guidelines. This filtration technique is vital for , thus bolstering the reliability of protein purification processes and the overall efficacy of biotechnological applications.
Food and Beverage Industry: Ensuring Safety with 0.22 Micron Filtration
In the food and beverage sector, the use of 0.22 micron screens is critical for ensuring the safety and quality of products. These advanced devices, like the syringe filter 0.22 micron, effectively eliminate microorganisms from beverages, such as juices and wines, thereby preventing spoilage and ensuring compliance with health regulations.
By integrating these enhancements into their manufacturing processes, producers not only improve product safety but also extend shelf life. This commitment to quality reflects a proactive approach to and regulatory adherence, reinforcing the industry's dedication to excellence.
Laboratory Research: Preparing Samples with 0.22 Micron Filters
Devices featuring a syringe filter 0.22 micron are essential tools in laboratory research, particularly for sample preparation. These devices use a syringe filter 0.22 micron to effectively from samples, thereby ensuring the accuracy and reliability of subsequent analyses. By utilizing these screens, researchers significantly mitigate contamination risks, which leads to enhanced quality in experimental outcomes. The importance of high-quality scientific instruments cannot be overstated; they are pivotal in achieving precise and trustworthy results in various research applications.
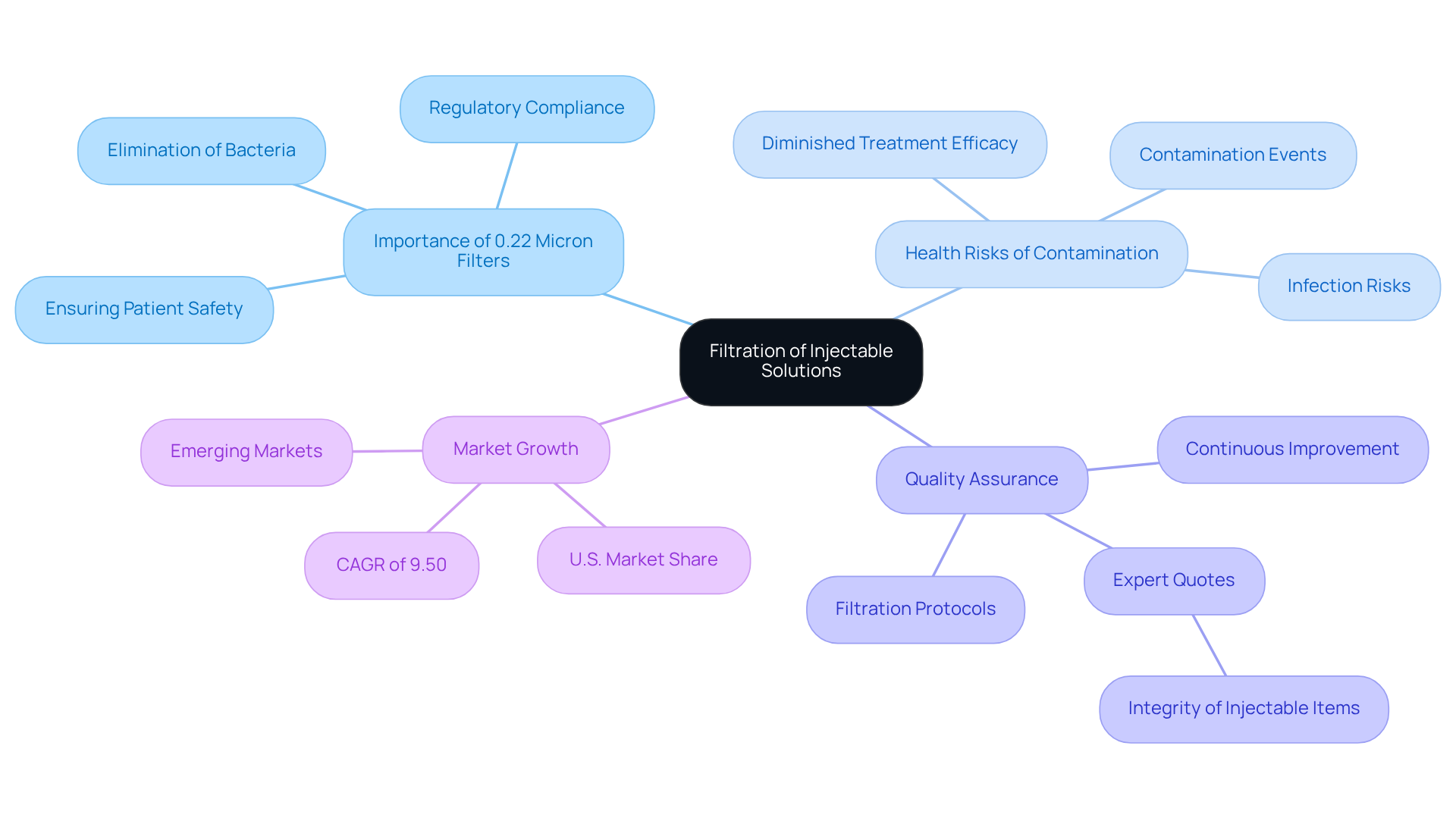
Pharmaceutical Industry: Filtration of Injectable Solutions Using 0.22 Micron Filters
In the pharmaceutical sector, syringe filter 0.22 micron screens play a vital role in the purification of injectable solutions. These filters effectively eliminate bacteria and other impurities, ensuring patient well-being. The necessity of such filtration is underscored by the serious health risks associated with contamination events in injectable products, which can lead to infections and diminished treatment efficacy. Research highlights that items lacking 0.22 micron filtration have faced significant contamination challenges, underscoring the critical need for rigorous filtration protocols.
Quality assurance experts emphasize the importance of maintaining safety standards. One expert noted, 'The integrity of injectable items relies on our filtration processes; without them, we jeopardize patient safety.' By adhering to stringent filtration methods, pharmaceutical companies not only comply with regulatory requirements but also uphold the highest standards of quality and safety in their injectable products. The proactive use of syringe filter 0.22 micron ensures the sterility of final formulations, ultimately enhancing patient outcomes and fostering trust in pharmaceutical therapies.
Moreover, the sterile filtration market is projected to grow at a CAGR of 9.50% from 2025 to 2032, reflecting an increasing recognition of the importance of effective filtration in maintaining product integrity. This anticipated growth demonstrates the industry's commitment to , particularly from sources such as water and steam, which can introduce impurities into the final product.
Clinical Laboratories: Filtering Biological Samples with 0.22 Micron Filters
In clinical laboratories, the application of 0.22-micrometer membranes is essential for purifying biological specimens, such as blood and urine, prior to analysis. These devices effectively eliminate impurities that could compromise , thereby ensuring precise and reliable outcomes. The use of 0.22-micrometer filtration significantly enhances diagnostic accuracy, which is crucial for optimal patient care.
Research indicates that approximately 1 in every 1,000 starved S. marcescens cells can pass through a 0.22-micrometer barrier, underscoring the importance of these membranes in mitigating the risk of microbial contamination in critical samples. Clinical lab professionals assert that implementing such filtration methods not only bolsters the integrity of test results but also facilitates improved clinical decision-making.
Furthermore, laboratory simulation testing has demonstrated that under certain conditions, bacteria can penetrate 0.2- and 0.22-μm membranes, highlighting the necessity of incorporating prefiltration stages to prevent microbial breakthrough. By utilizing a syringe filter 0.22 micron, laboratories can uphold high standards of quality in diagnostics, ultimately leading to enhanced patient outcomes.
Research and Development: Formulating New Products with 0.22 Micron Filters
In the realm of research and development, 0.22-micrometer screens are indispensable in the creation of new products, ensuring that components remain free from impurities. This aspect is particularly crucial in the pharmaceutical and biotechnology sectors, where the maintenance of purity is vital for both efficacy and safety.
Research indicates that without the implementation of 0.22-micron screens, contamination levels in new formulations can significantly increase, leading to compromised quality and potential regulatory issues. By utilizing these filters, researchers can effectively eliminate bacteria, fungi, and other microorganisms, thus preserving the integrity of their formulations.
R&D experts assert that filtration at a size of 0.22 micrometers not only enhances product stability but also adheres to stringent industry standards for sterility. This unwavering commitment to is essential for successful product development, especially in the formulation of injectable drugs and sensitive biopharmaceuticals.
Best Practices: Handling and Maintaining 0.22 Micron Filters in Laboratories
To ensure optimal performance of the syringe filter 0.22 micron, it is crucial to adhere to best practices for handling and maintenance.
- Employing sterile techniques during filtration is essential to prevent contamination.
- Avoiding excessive pressure is vital, as it can lead to damage and performance degradation.
- Furthermore, storing screens in a clean, dry environment is necessary; moisture and contaminants can jeopardize their integrity.
- Regular inspections for physical damage and strict adherence to expiration dates are vital for maintaining effectiveness in laboratory applications.
Laboratory managers emphasize that neglecting these maintenance practices can result in significant performance issues, including reduced filtration efficiency and an increased risk of contamination.
For instance, maintaining the performance of 0.22 micron membranes often requires the use of a syringe filter 0.22 micron in laboratory settings, alongside routine checks and proper storage protocols, which are critical for ensuring reliable results in sensitive experiments.
Ultimately, not only prolongs the life of the filters but also enhances the overall reliability of laboratory processes.
Conclusion
The significance of 0.22 micron syringe filters in laboratory settings is paramount. These filters serve as indispensable tools that guarantee the purity and reliability of samples across diverse applications, ranging from pharmaceutical production to environmental testing and biotechnology. Their efficacy in eliminating microorganisms and contaminants positions them as vital components in upholding high standards of safety and quality in scientific research.
Throughout this discussion, the key applications of 0.22 micron filters have been clearly outlined, including their essential roles in:
- Vaccine manufacturing
- Clinical diagnostics
- Food safety
The emphasis on the filters' contributions to regulatory compliance, product integrity, and enhanced research outcomes underscores their critical nature. Moreover, the importance of adhering to best practices for the handling and maintenance of these filters has been highlighted, as proper care directly influences their performance and the reliability of laboratory results.
Given the increasing demand for precise sample purification and the anticipated growth of the syringe membrane market, it is imperative for laboratories to acknowledge the value of 0.22 micron filters. By integrating these filters into standard operating procedures, researchers and technicians can ensure the accuracy of their findings and uphold the safety of their products. Embracing these filtration technologies not only enhances scientific integrity but also contributes to the advancement of health and safety standards across various industries.




